The correct answer is Option (3) →
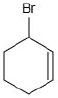
An allylic halide is a compound in which the halogen atom is attached to a carbon next to a double bond (C = C), not directly on the double-bond carbon.
Reasoning for each option:
Option 1 – Bromobenzene type structure
Here bromine is directly attached to a benzene ring carbon.
This is an aryl halide, not allylic.
Option 2 – Bromine on the double-bond carbon of cyclohexene
Halogen is attached directly to the C = C carbon.
This is a vinylic halide, not allylic.
Option 3 – Bromine on the carbon adjacent to the double bond
Bromine is on the carbon next to the double bond.
This matches the definition of an allylic halide.
Option 4 – CH₂ = CHBr
Bromine is directly bonded to a double-bond carbon.
This is also a vinylic halide, not allylic.
|