The correct answer is Option (4) → (B), (C) and (D) only
(A) Phenol to Salicylic Acid (o-Hydroxybenzoic Acid)
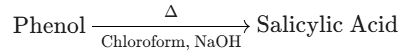
- Intended Product: Salicylic Acid (o-Hydroxybenzoic Acid), which has a –COOH group at the ortho position.
- Reagents: Chloroform ($CHCl_3$) and NaOH.
- Actual Reaction: Phenol with $CHCl_3$ and NaOH is the Reimer-Tiemann Reaction, which introduces an aldehyde (–CHO) group, yielding Salicylaldehyde (o-Hydroxybenzaldehyde).
- Conclusion: The reagents shown ($CHCl_3$, NaOH) do not produce the –COOH group (Salicylic Acid). The correct reagents for Salicylic Acid are $CO_2$ and NaOH (Kolbe's Reaction). Therefore, conversion (A) is Incorrect.
(B) Phenol to p-Benzoquinone
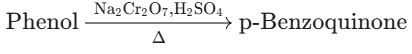
- Reaction Type: Oxidation of Phenol.
- Reagents: Sodium dichromate ($Na_2Cr_2O_7$) in an acidic medium ($H_2SO_4$) is a strong oxidizing agent.
- Product: Phenol is oxidized to p−Benzoquinone. * Conclusion: The conversion of Phenol to p−Benzoquinone using this strong oxidizing agent is Correct.
(C) Phenol to Salicylaldehyde (o-Hydroxybenzaldehyde)
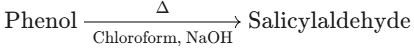
- Reaction Type: Reimer-Tiemann Reaction.
- Reagents: Phenol reacts with Chloroform ($CHCl_3$) in the presence of an aqueous base (NaOH).
- Product: The reaction introduces an aldehyde (–CHO) group at the ortho position, yielding Salicylaldehyde.
- Conclusion: The conversion of Phenol to Salicylaldehyde is Correct.
(D) Phenol to Benzene
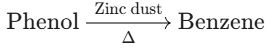
- Reaction Type: Reduction of Phenol.
- Reagents: Phenol is heated (distilled) with Zinc dust (Zn).
- Product: Zinc reduces the hydroxyl (–OH) group, forming Benzene ($C_6H_6$) and Zinc Oxide (ZnO). * Conclusion: The conversion of Phenol to Benzene using Zinc dust is Correct.
Summary
The correct conversions are (B), (C), and (D). |