The correct answer is Option (1) →
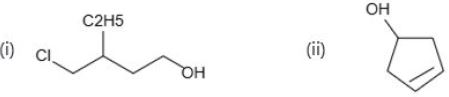
Based on the IUPAC names provided in the image, the correct structures are found in Option 1.
Here is the step-by-step breakdown of how to identify these structures:
(i) 4-Chloro-3-ethylbutan-1-ol
To find this structure, let's deconstruct the name:
- Parent chain: "butan-1-ol" means a 4-carbon chain with an alcohol group ($-OH$) on carbon 1.
- Substituents: An ethyl group ($-C_{2}H_{5}$) on carbon 3 and a chlorine atom ($-Cl$) on carbon 4.
Analysis of the options:
- Option 1: Carbon 1 has the $-OH$. Carbon 3 has the ethyl group. Carbon 4 has the $Cl$. This matches perfectly.
- Option 2: This shows a 5-carbon parent chain (pentan-1-ol), which is incorrect.
- Option 3: The $-OH$ is on carbon 2, making it a "butan-2-ol" derivative.
- Option 4: This is a 1-chlorobutan-2-ol derivative.
(ii) Cyclopent-3-en-1-ol
- Parent ring: "Cyclopent" means a 5-carbon ring.
- Alcohol: "-1-ol" means the carbon attached to the $-OH$ is designated as carbon 1.
- Alkene: "-3-en" means the double bond starts at carbon 3.
Analysis of the options:
- Option 1: Starting from the $-OH$ as carbon 1 and counting towards the double bond, the double bond begins at carbon 3. This is correct.
- Option 2: The double bond is at carbon 2 (Cyclopent-2-en-1-ol).
- Option 3: This is an enol (the double bond is on the same carbon as the $-OH$).
- Option 4: Similar to Option 2, the double bond is in the 2nd position relative to the alcohol.
|