The correct answer is Option (2) →
A vinylic halide is a compound in which the halogen atom is directly attached to a carbon that is part of a double bond (C = C).
Rule:
-
The halogen must be on the double-bond carbon itself.
-
If the halogen is on a benzene ring, it is an aryl halide, not vinylic.
-
If the halogen is on the carbon next to a double bond, it is an allylic halide, not vinylic.
Option 1 – C₂H₅X
This is an alkyl halide. There is no double bond.
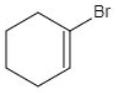
Option 2 – Cyclohexene with Br on the double-bond carbon
Bromine is directly attached to the carbon involved in the double bond. This fits the definition of a vinylic halide.
Option 3 – Bromobenzene
Halogen is attached to a benzene ring. This is an aryl halide, not vinylic.
Option 4 – Chlorine on a single-bond carbon of cyclohexene
Halogen is not on the double-bond carbon. This is an allylic halide.
|