The correct answer is OPTION 1 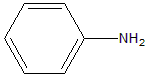
Option 1 (Aniline, –NH₂ directly attached to benzene) has the highest ( pK_b ) value.
( pK_b ) is inversely related to basic strength. Higher ( pK_b ) means weaker base.
-
In aniline (Option 1), the lone pair on nitrogen is delocalised into the benzene ring by resonance, making it least available for protonation → weakest base → highest ( pK_b )
-
In benzylamine (Option 2), the –NH₂ is not directly attached to the ring, so no resonance → strong base
-
In N-methylaniline (Option 3) and N,N-dimethylaniline (Option 4), +I effect of alkyl groups increases electron density → stronger than aniline
Therefore, the weakest base (highest ( pK_b )) is: Option (1) |