The correct answer is Option (3) →
Reason (step-by-step):
Now compare the given compounds:
-
Benzoic acid (C₆H₅COOH)
– Phenyl ring shows a –I effect, stabilising the conjugate base
– Hence more acidic → lower pKₐ
-
Phenylacetic acid (C₆H₅CH₂COOH)
– Phenyl group still exerts a –I effect, though weaker than benzoic acid
– Still more acidic than acetic acid
-
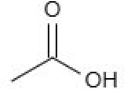
Acetic acid (CH₃COOH)
– CH₃ group has +I effect, which destabilises the conjugate base
– Hence weakest acid → highest pKₐ
-
Chloro-substituted acid (Cl–CH₂–CH₂–COOH)
– Cl has strong –I effect, greatly stabilising conjugate base
– Therefore most acidic → lowest pKₐ
Final order of acidity (strong → weak):
Chloro acid > Benzoic acid > Phenylacetic acid > Acetic acid
Thus, the compound with the highest pKₐ is:
Acetic acid (Option 3) |