The correct answer is Option (3) → (B), (A), (D), (C)
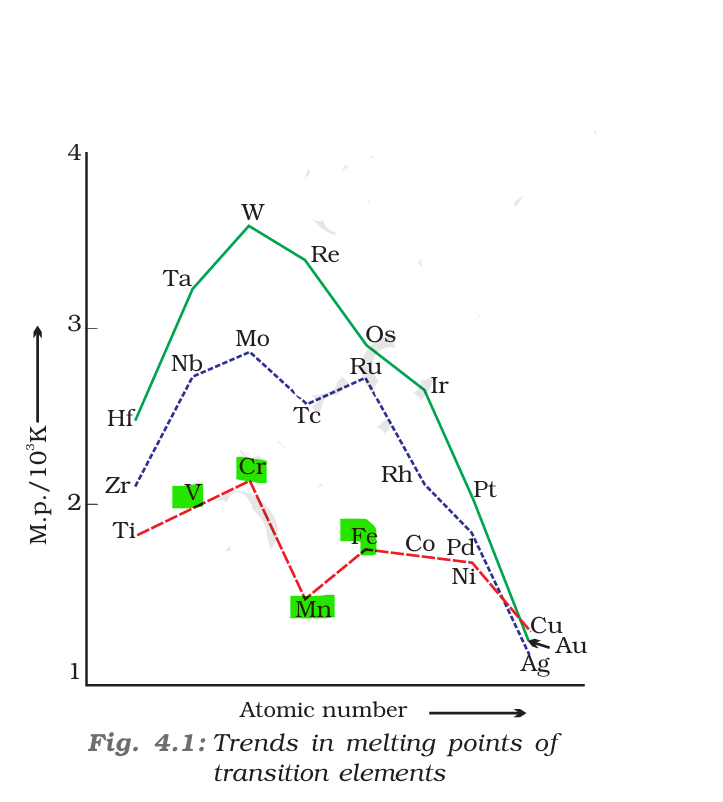
Analysis of the Metals
- Chromium (Cr): Chromium has a $3d^5 4s^1$ configuration. With six unpaired electrons available for bonding, it has a very high melting point (~1907°C).
- Vanadium (V): Vanadium has a $3d^3 4s^2$ configuration. It possesses strong metallic bonding, resulting in a high melting point (~1910°C), though in many periodic trends, it sits very close to Chromium.
- Iron (Fe): Iron has a $3d^6 4s^2$ configuration. While it has strong metallic bonds, the melting point (~1538°C) is lower than that of Chromium or Vanadium.
- Manganese (Mn): Manganese is a notable anomaly in the first transition series. Despite having a $3d^5 4s^2$ configuration (half-filled d-subshell), the electrons are held more tightly, and it has a complex crystal structure that results in relatively weak metallic bonding. Consequently, it has a significantly lower melting point (~1246°C) compared to its neighbors.
The increasing order is: Mn (B) < Fe (A) < V (D) < Cr (C).
|