The correct answer is Option (1) →
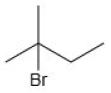
(tertiary alkyl bromide)
An SN1 reaction proceeds via carbocation formation. The rate depends mainly on carbocation stability:
Tertiary > Secondary > Primary
-
Option 1 forms a tertiary carbocation, which is most stable due to hyperconjugation and +I effect → fastest SN1
-
Options 2 and 3 form secondary carbocations
-
Option 4 forms a primary carbocation, least stable → slowest
|